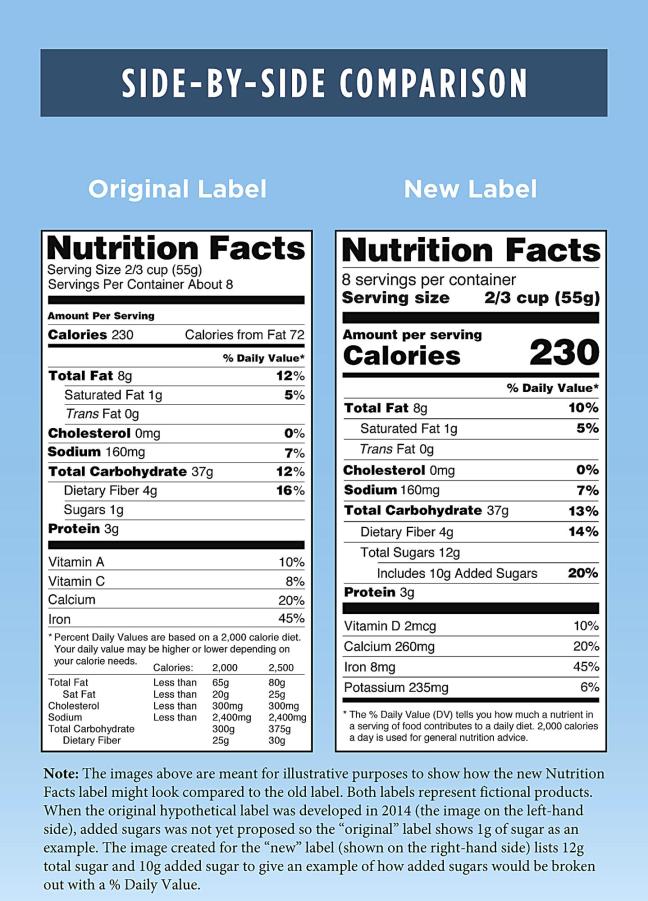
On 3rd May 2018 the U.S. Food and Drug Administration has issued a final rule to extend the compliance dates for updating Nutrition Facts and Supplement Facts labels, from July 26, 2018, to January 1, 2020, for manufacturers with $10 million or more in annual food sales. Manufacturers with less than $10 million in annual food sales … Continue reading FDA finalizes the extension of the compliance dates for new Nutrition Facts
FDA finalizes the extension of the compliance dates for new Nutrition Facts